Can 70% Alcohol Be Used To Clean Up Vincristine
Editor-In-Principal: C. Michael Gibson, M.South., M.D. [one]; Associate Editor(s)-in-Main: Gloria Picoy [2];Aparna Vuppala, Thou.B.B.S. [3]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, non a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practise guidelines / consensus statements. WikiDoc does not promote the assistants of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
Warnings
See full prescribing information for consummate Boxed Alert.
Caution-This grooming should be administered by individuals experienced in the administration of vincristine sulfate. It is extremely important that the intravenous needle or catheter be properly positioned before any vincristine is injected. Leakage into surrounding tissue during intravenous administration of vincristine sulfate may cause considerable irritation. If extravasation occurs, the injection should be discontinued immediately, and any remaining portion of the dose should then exist introduced into another vein. Local injection of hyaluronidase and the application of moderate heat to the area of leakage help disperse the drug and are thought to minimize discomfort and the possibility of cellulitis. FOR INTRAVENOUS USE Just – FATAL IF GIVEN BY OTHER ROUTES.
Overview
Vincristine sulfate is a mitotic inhibitor that is FDA approved for the treatment of astute leukemia, Hodgkin's disease, non-Hodgkin'south cancerous lymphomas, rhabdomyosarcoma, neuroblastoma, Wilms' tumor. In that location is a Black Box Warning for this drug equally shown here . Common agin reactions include baldness, constipation, nausea and airsickness.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Vincristine sulfate is indicated in acute leukemia. Has also been shown to be useful in combination with other oncolytic agents in Hodgkin'due south disease, non-Hodgkin'south malignant lymphomas, rhabdomyosarcoma, neuroblastoma, and Wilms' tumor.
- Dosage: 1.iv mg/m2 at weekly intervals.
Off-Label Apply and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Vincristine sulfate in developed patients.
Non–Guideline-Supported Use
- AIDS-related Kaposi's sarcoma
-
- Dosage: two mg on odd numbered weeks [1]
- Blastic stage chronic myeloid leukemia
- Chest cancer
- Cervical cancer
- Chronic lymphoid leukemia
- Colorectal cancer
- Ewing's sarcoma of bone
- Germ prison cell tumor of ovary
- Gestational trophoblastic neoplasia
- Head and neck cancer
- Hepatoblastoma
- Idiopathic thrombocytopenic purpura
- Intracranial tumor
- Kaposi'due south sarcoma
- Malignant epithelial tumor of ovary
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Dosage: ane.v to ii mg/m2
- Dosage for pediatric patients weighing ten kg or less, the starting dose should be 0.05 mg/kg, administered once a week.
Off-Label Employ and Dosage (Pediatric)
Guideline-Supported Apply
In that location is express information regarding Off-Label Guideline-Supported Use of Vincristine sulfate in pediatric patients.
Non–Guideline-Supported Use
- Ewing's sarcoma of bone
- Germ prison cell tumor of ovary
Contraindications
- Demyelinating grade of Charcot-Marie-Tooth syndrome
Warnings
Warnings
Run across full prescribing data for consummate Boxed Alarm.
Caution-This preparation should be administered past individuals experienced in the assistants of vincristine sulfate. It is extremely important that the intravenous needle or catheter be properly positioned before whatever vincristine is injected. Leakage into surrounding tissue during intravenous assistants of vincristine sulfate may cause considerable irritation. If extravasation occurs, the injection should be discontinued immediately, and whatever remaining portion of the dose should then exist introduced into some other vein. Local injection of hyaluronidase and the application of moderate heat to the surface area of leakage help disperse the drug and are thought to minimize discomfort and the possibility of cellulitis. FOR INTRAVENOUS USE ONLY – FATAL IF GIVEN BY OTHER ROUTES.
- This grooming is for intravenous utilize merely. It should be administered past individuals experienced in the administration of vincristine sulfate injection. The intrathecal assistants of vincristine sulfate injection usually results in death.
- To reduce the potential for fatal medication errors due to wrong road of assistants, vincristine sulfate should be diluted in a flexible plastic container and prominently labeled every bit indicated for intravenous use only.
- Syringes containing this product must be labeled, using the auxiliary sticker provided, to state "FOR INTRAVENOUS USE ONLY – FATAL IF GIVEN Past OTHER ROUTES."
- Extemporaneously prepared syringes containing this product must be packaged in an overwrap which is labeled "practise non remove roofing until moment of injection. For intravenous employ only – fatal if given by other routes."
Adverse Reactions
Clinical Trials Experience
- In general, adverse reactions are reversible and are related to dosage. The most common agin reaction is pilus loss; the most troublesome adverse reactions are neuromuscular in origin.
- When unmarried, weekly doses of the drug are employed, the agin reactions of leukopenia, neuritic pain, and constipation occur just are usually of short duration (i.e., less than 7 days). When the dosage is reduced, these reactions may lessen or disappear. The severity of such reactions seems to increment when the calculated amount of drug is given in divided doses.
- Other adverse reactions, such every bit pilus loss, sensory loss, paresthesia, difficulty in walking, slapping gait, loss of deep-tendon reflexes, and musculus wasting, may persist for at least as long as therapy is connected. Generalized sensorimotor dysfunction may become progressively more astringent with continued treatment. Although most such symptoms unremarkably disappear by about the 6th week after discontinuance of treatment, some neuromuscular difficulties may persist for prolonged periods in some patients. Regrowth of hair may occur while maintenance therapy continues.
The following agin reactions take been reported:
Hepatic veno-occlusive disease
- It has been reported in patients receiving vincristine, particularly in pediatric patients, as function of standard combination chemotherapy regimens. Some of the patients had fatal outcomes; some who survived had undergone liver transplantation.
Hypersensitivity
- Rare cases of allergic-blazon reactions, such as anaphylaxis, rash, and edema, that are temporally related to vincristine therapy have been reported in patients receiving vincristine every bit a part of multidrug chemotherapy regimens.
Gastrointestinal
- Constipation, intestinal cramps, weight loss, nausea, airsickness, oral ulceration, diarrhea, paralytic ileus, intestinal necrosis and/or perforation, and anorexia have occurred.
- Constipation may accept the form of upper-colon impaction, and, on physical exam, the rectum may be empty. Colicky abdominal pain coupled with an empty rectum may mislead the dr.. A flat film of the belly is useful in demonstrating this condition. All cases have responded to high enemas and laxatives. A routine prophylactic regimen against constipation is recommended for all patients receiving vincristine sulfate.
- Paralytic ileus may occur, peculiarly in immature pediatric patients. The ileus volition reverse itself with temporary discontinuance of vincristine sulfate and with symptomatic intendance.
Genitourinary
- Polyuria, dysuria, and urinary retention due to bladder atony take occurred. Other drugs known to crusade urinary retention (specially in the elderly) should, if possible, be discontinued for the starting time few days following administration of vincristine sulfate.
Cardiovascular
- Hypertension and hypotension take occurred. Chemotherapy combinations that have included vincristine sulfate, when given to patients previously treated with mediastinal radiation, have been associated with coronary avenue disease and myocardial infarction. Causality has not been established.
Neurologic
- Often, there is a sequence to the development of neuromuscular side furnishings.
- Initially, only sensory impairment and paresthesia may exist encountered. With connected treatment, neuritic hurting and, later, motor difficulties may occur. There have been no reports of whatever agent that can reverse the neuromuscular manifestations that may accompany therapy with vincristine sulfate.
- Loss of deep-tendon reflexes, foot drop, ataxia, and paralysis accept been reported with continued administration. Cranial nerve manifestations, such as isolated paresis and/or paralysis of muscles controlled by cranial motor fretfulness including potentially life-threatening bilateral vocal string paralysis, may occur in the absence of motor harm elsewhere; extraocular and laryngeal muscles are those most unremarkably involved. Jaw pain, pharyngeal pain, parotid gland pain, bone pain, dorsum hurting, limb pain, and myalgias take been reported; hurting in these areas may exist severe.
- Convulsions, frequently with hypertension, have been reported in a few patients receiving vincristine sulfate. Several instances of convulsions followed by coma have been reported in pediatric patients.
- Transient cortical blindness and optic cloudburst with blindness
- Treatment with vinca alkaloids has resulted in both vestibular and auditory damage to the eighth cranial nerve.
- Manifestations include partial or total deafness which may exist temporary or permanent, and difficulties with balance including dizziness, nystagmus, and vertigo.
Item caution is warranted when vincristine is used in combination with other agents known to be ototoxic such every bit the platinum-containing oncolytics.
Pulmonary
- Astute shortness of jiff and astringent bronchospasm accept been reported following the administration of vinca alkaloids. These reactions accept been encountered most frequently when the vinca alkaloid was used in combination with mitomycin-C and may require ambitious treatment, particularly when there is preexisting pulmonary dysfunction. The onset of these reactions may occur minutes to several hours after the vinca alkaloid is injected and may occur upwards to 2 weeks following the dose of mitomycin.
- Progressive dyspnea requiring chronic therapy may occur. Vincristine sulfate should non be readministered.
Endocrine
- Rare occurrences of a syndrome attributable to inappropriate antidiuretic hormone secretion take been observed in patients treated with vincristine sulfate.
Hematologic
Vincristine sulfate does not appear to have any constant or significant effect on platelets or ruddy claret cells.
- Serious os-marrow depression is unremarkably not a major dose-limiting event.
- Anemia, leukopenia, and thrombocytopenia accept been reported.
- Thrombocytopenia, if present when therapy with vincristine sulfate is begun, may actually ameliorate earlier the advent of os marrow remission.
Pare
- Alopecia
- Rash
Other
- Fever
- Headache
Postmarketing Feel
At that place is limited information regarding Vincristine sulfate Postmarketing Experience in the drug label.
Drug Interactions
The simultaneous oral or intravenous assistants of phenytoin and antineoplastic chemotherapy combinations that included vincristine sulfate has been reported to reduce blood levels of the anticonvulsant and to increment seizure activity. Dosage adjustment should exist based on serial claret level monitoring. The contribution of vincristine sulfate to this interaction is not certain. The interaction may consequence from reduced absorption of phenytoin and an increment in the rate of its metabolism and emptying.
Caution should be exercised in patients concurrently taking drugs known to inhibit drug metabolism by hepatic cytochrome P450 isoenzymes in the CYP 3A subfamily, or in patients with hepatic dysfunction. Concurrent administration of vincristine sulfate with itraconazole (a known inhibitor of the metabolic pathway) has been reported to cause an earlier onset and/or an increased severity of neuromuscular side effects. This interaction is presumed to be related to inhibition of the metabolism of vincristine.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): D Vincristine sulfate can cause fetal harm when administered to a meaning woman. When significant mice and hamsters were given doses of vincristine sulfate that caused resorption of 23% to 85% of fetuses, fetal malformations were produced in those that survived. 5 monkeys were given single doses of vincristine sulfate between days 27 and 34 of their pregnancies; iii of the fetuses were normal at term, and 2 viable fetuses had grossly evident malformations at term. In several animal species, vincristine sulfate can induce teratogenesis as well equally embryo death at doses that are nontoxic to the meaning fauna. There are no adequate and well-controlled studies in meaning women. If this drug is used during pregnancy or if the patient becomes significant while receiving this drug, she should be apprised of the potential hazard to the fetus. Women of childbearing potential should exist advised to avert condign pregnant.
Pregnancy Category (AUS): D There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Vincristine sulfate in women who are meaning.
Labor and Commitment
At that place is no FDA guidance on use of Vincristine sulfate during labor and delivery.
Nursing Mothers
It is non known whether this drug is excreted in human milk. Considering many drugs are excreted in bhuman milk and considering of the potential for serious adverse reactions due to vincristine sulfate in nursing infants, a conclusion should be made either to discontinue nursing or the drug, taking into business relationship the importance of the drug to the female parent.
Pediatric Apply
There is no FDA guidance on the use of Vincristine sulfate in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Vincristine sulfate in geriatric settings.
Gender
At that place is no FDA guidance on the use of Vincristine sulfate with respect to specific gender populations.
Race
There is no FDA guidance on the utilise of Vincristine sulfate with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the utilise of Vincristine sulfate in patients with renal harm.
Hepatic Harm
There is no FDA guidance on the use of Vincristine sulfate in patients with hepatic impairment.
Females of Reproductive Potential and Males
Neither in vivo nor in vitro laboratory tests accept conclusively demonstrated the mutagenicity of this product. Fertility following treatment with vincristine sulfate alone for cancerous disease has non been studied in humans. Clinical reports of both male and female patients who received multiple-agent chemotherapy that included vincristine sulfate signal that azoospermia and amenorrhea can occur in postpubertal patients. Recovery occurred many months afterward completion of chemotherapy in some but not all patients. When the same handling is administered to prepubertal patients, permanent azoospermia and amenorrhea are much less likely.
Patients who received chemotherapy with vincristine sulfate in combination with anti-cancer drugs known to be carcinogenic have developed second malignancies. The contributing role of vincristine sulfate in this evolution has non been determined. No bear witness of carcinogenicity was found following intraperitoneal administration of vincristine sulfate in rats and mice, although this study was limited.
Immunocompromised Patients
There is no FDA guidance 1 the use of Vincristine sulfate in patients who are immunocompromised.
Administration and Monitoring
Assistants
Intravenous
Monitoring
There is limited information regarding Vincristine sulfate Monitoring in the drug characterization.
IV Compatibility
There is limited information regarding the compatibility of Vincristine sulfate and IV administrations.
Overdosage
Side furnishings following the use of vincristine sulfate are dose related.
- In pediatric patients under 13 years of age, death has occurred post-obit doses of vincristine sulfate that were x times those recommended for therapy. Astringent symptoms may occur in this patient group post-obit dosages of 3 to 4 mg/m2.
- Adults tin be expected to experience severe symptoms later on single doses of three mg/m2 or more. Therefore, post-obit administration of doses college than those recommended, patients can be expected to experience exaggerated side effects. Supportive care should include the following:
-
- Prevention of side effects resulting from the syndrome of inappropriate antidiuretic hormone secretion (preventive treatment would include restriction of fluid intake and perhaps the administration of a diuretic affecting the part of Henle'due south loop and the distal tubule)
- Administration of anticonvulsants
- Utilise of enemas or cathartics to preclude ileus (in some instances, decompression of the gastrointestinal tract may be necessary)
- Monitoring the cardiovascular system
- Determining daily blood counts for guidance in transfusion requirements.
Folinic acid has been observed to have a protective effect in normal mice that were administered lethal doses of vincristine sulfate. Isolated instance reports propose that folinic acid may be helpful in treating humans who have received an overdose of vincristine sulfate. It is suggested that 100 mg of folinic acid be administered intravenously every 3 hours for 24 hours and then every half dozen hours for at least 48 hours. Theoretically (based on pharmacokinetic information), tissue levels of vincristine sulfate can exist expected to remain significantly elevated for at to the lowest degree 72 hours. Treatment with folinic acid does not eliminate the demand for the above-mentioned supportive measures.
Most of an intravenous dose of vincristine is excreted into the bile later rapid tissue bounden. Considering only very minor amounts of the drug appear in dialysate, hemodialysis is not likely to be helpful in cases of overdosage. An increase in the severity of side effects may be experienced past patients with liver disease that is astringent enough to decrease biliary excretion.
Enhanced fecal excretion of parenterally administered vincristine has been demonstrated in dogs pretreated with cholestyramine. In that location are no published clinical data on the utilise of cholestyramine as an antidote in humans.
At that place are no published clinical information on the consequences of oral ingestion of vincristine. Should oral ingestion occur, the breadbasket should be evacuated. Evacuation should be followed by oral assistants of activated charcoal and a cathartic.
Treatment of patients following intrathecal administration of vincristine sulfate injection has included immediate removal of spinal fluid and flushing with Lactated Ringer'southward, likewise as other solutions and has not prevented ascending paralysis and death. In 1 case, progressive paralysis in an developed was arrested by the following treatment initiated immediately after the intrathecal injection:
- Every bit much spinal fluid was removed as could be safely washed through lumbar access.
- The subarachnoid infinite was flushed with Lactated Ringer's solution infused continuously through a catheter in a cerebral lateral ventricle at the rate of 150 mL/h. The fluid was removed through a lumbar access.
- Equally soon as fresh frozen plasma became bachelor, the fresh frozen plasma, 25 mL, diluted in 1 Fifty of Lactated Ringer's solution was infused through the cognitive ventricular catheter at the rate of 75 mL/h with removal through the lumbar admission. The rate of infusion was adjusted to maintain a protein level in the spinal fluid of 150 mg/dL.
- Glutamic acid, 10 g, was given intravenously over 24 hours followed by 500 mg three times daily by mouth for i month or until neurological dysfunction stabilized. The role of glutamic acrid in this handling is not sure and may not be essential.
Pharmacology
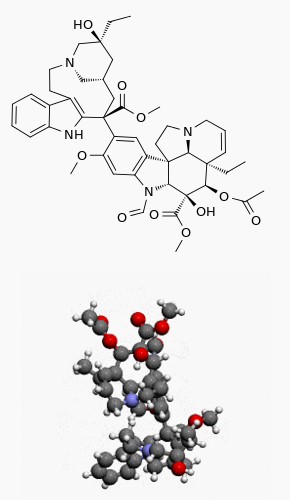 | |
| Vincristine sulfate | |
| Systematic (IUPAC) name | |
| (3aR,3a1R,4R,5S,5aR,10bR)-methyl 4-acetoxy-3a-ethyl-9-((5S,7S,9S)-5-ethyl-five-hydroxy-9-(methoxycarbonyl)-2,4,5,6,7,8,9,10-octahydro-1H-3,vii-methano[i]azacycloundecino[5,4-b]indol-9-yl)-6-formyl-5-hydroxy-8-methoxy-3a,3a1,4,5,5a,vi,11,12-octahydro-1H-indolizino[eight,one-cd]carbazole-5-carboxylate | |
| Identifiers | |
| CAS number | |
| ATC code | L01 |
| PubChem | |
| DrugBank | |
| Chemic information | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 824.958 g/mol |
| SMILES | & |
| Pharmacokinetic information | |
| Bioavailability | north/a (not reliably absorbed past the GI tract) |
| Protein bounden | ~44%[2] |
| Metabolism | Liver, mostly via CYP3A4 and CYP3A5 |
| Half life | 19 to 155 hours (mean: 85 hours) |
| Excretion | Faeces (70-80%), urine (x-20%) |
| Therapeutic considerations | |
| Pregnancy cat. | D(AU) D(United states) |
| Legal status | Prescription Only (S4)(AU) ?(CA) POM(Uk) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | intravenous |
Mechanism of Action
The mechanisms of activeness of vincristine sulfate remain under investigation. The mechanism of activity of vincristine sulfate has been related to the inhibition of microtubule formation in mitotic spindle, resulting in an arrest of dividing cells at the metaphase phase.
Structure
The structural formula is as follows:

Vincristine Sulfate, USP is a white to slightly yellow pulverisation. It is soluble in methanol, freely soluble in water, but only slightly soluble in 95% ethanol. In 98% ethanol, vincristine sulfate, USP has an ultraviolet spectrum with maxima at 221 nm (∈ +47,100).
Pharmacodynamics
At that place is limited information regarding pharmacodynamics of vincristine sulfate
Pharmacokinetics
Pharmacokinetic studies in patients with cancer have shown a triphasic serum decay design following rapid intravenous injection. The initial, centre, and last one-half-lives are 5 minutes, 2.three hours, and 85 hours respectively; however, the range of the terminal half-life in humans is from 19 to 155 hours. The liver is the major excretory organ in humans and animals. The metabolism of vinca alkaloids has been shown to exist mediated by hepatic cytochrome P450 isoenzymes in the CYP 3A subfamily. This metabolic pathway may exist impaired in patients with hepatic dysfunction or who are taking concomitant potent inhibitors of these isoenzymes . About fourscore% of an injected dose of vincristine sulfate appears in the carrion and 10% to 20% tin exist found in the urine. Within xv to xxx minutes after injection, over 90% of the drug is distributed from the blood into tissue, where it remains tightly, but not irreversibly, spring.
Nonclinical Toxicology
In that location is limited information regarding Vincristine sulfate Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Vincristine sulfate Clinical Studies in the drug characterization.
How Supplied
Vincristine sulfate injection 1 mg/mL Unmarried Apply Vial
- NDC 0703-4402-xi
Vincristine sulfate injection two mg/2 mL Single Use Vial
- NDC 0703-4412-11
Storage
Shop under refrigeration between ii° to 8°C (36° to 46°F).
Images
Drug Images
Package and Label Display Panel

| |
| This paradigm of the FDA characterization is provided by the National Library of Medicine. |
| |
| This prototype of the FDA label is provided past the National Library of Medicine. |
Patient Counseling Data
There is limited data regarding Vincristine sulfate Patient Counseling Information in the drug characterization.
Precautions with Alcohol
Alcohol-Vincristine sulfate interaction has non been established. Talk to your doctor near the effects of taking alcohol with this medication.
Brand Names
- Oncovin
- Vincasar PFS [three]
Look-Alike Drug Names
At that place is express information regarding Vincristine sulfate Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA characterization are provided by the National Library of Medicine.
- ↑ Kaplan 50, Abrams D, Volberding P (1986). "Treatment of Kaposi's sarcoma in caused immunodeficiency syndrome with an alternating vincristine-vinblastine regimen". Cancer Care for Rep. lxx (ix): 1121–2. PMID 3742492.
- ↑ "Oncovin, Vincasar PFS (vincristine) dosing, indications, interactions, adverse furnishings, and more". Medscape Reference. WebMD. Retrieved 16 April 2022.
- ↑ "FDA LABEL: VINCASAR PFS- vincristine sulfate injection, solution".
Source: https://www.wikidoc.org/index.php/Vincristine_sulfate
Posted by: stephensonfriter54.blogspot.com




0 Response to "Can 70% Alcohol Be Used To Clean Up Vincristine"
Post a Comment